Identification of chiral carbon explains whether a molecule is chiral or achiral. Why is it Important to Identify Chiral Carbons
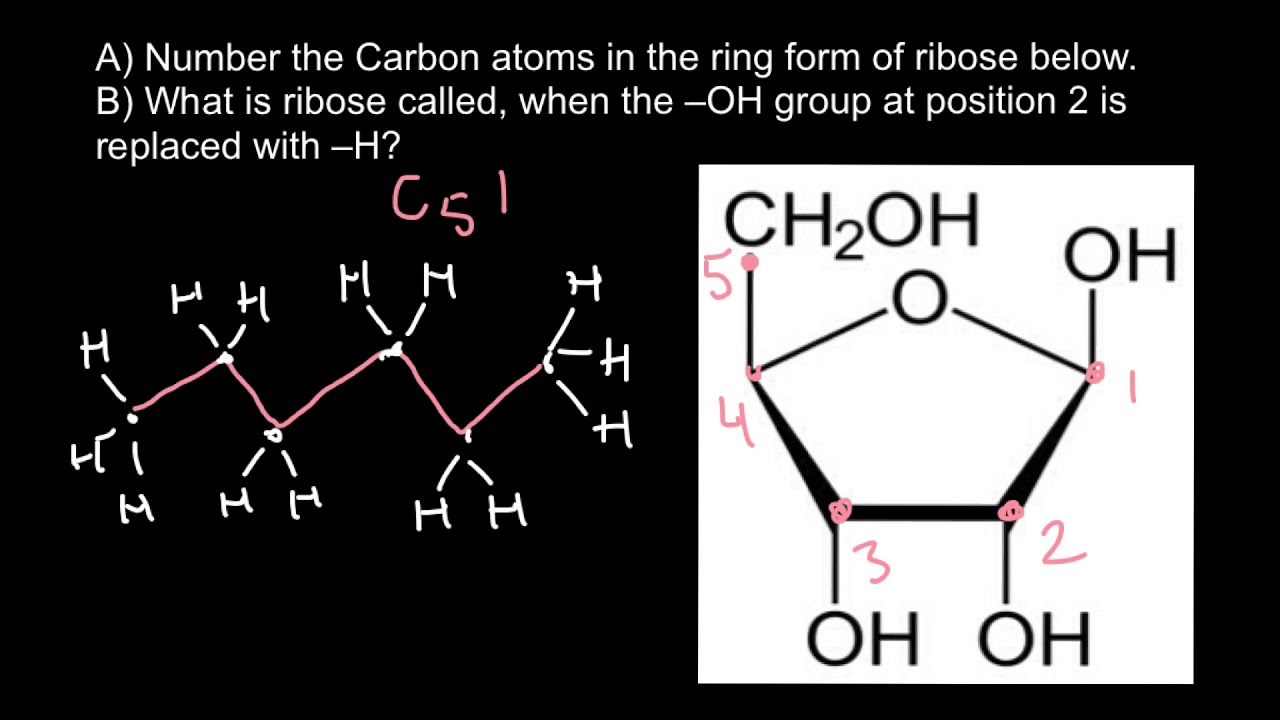
Therefore, the assumed carbon atom is achiral. When considering the above molecule, both hypothetical ligands are identical because there are no other substitutions in the ring structure. Now we can determine whether the assumed chiral carbon atom is chiral or achiral. Then break the adjacent bond on the right side and imagine it as the other ligand. The easiest way to do that is breaking the bond between the atom that is assumed to be the chiral carbon and the adjacent carbon atom on the left side and imagine it as a ligand. Step 2Ĭonvert the two groups in the ring into hypothetical groups which are not in a ring.

In the above image, the molecule has a hydrogen atom and a methyl group attached to the same carbon atom. If they are different, we can guess that it as a chiral carbon. – If the carbon atom with the tetrahedral arrangement is attached to four different groups, it is a chiral carbon.įirst, determine whether the groups attached to the carbon atom are different from each other. Step 2ĭetermine whether the four groups attached to that carbon atom are different from each other. If the geometry is not tetrahedral, then it is achiral. – If the geometry around the carbon atom is tetrahedral, then it can be a chiral carbon. Step 1ĭetermine the geometry of the molecule, taking the atom which is assumed to be the chiral carbon in the center.
#CHEMDRAW NUMBER CARBONS HOW TO#
How to Identify Chiral Carbons How to Identify Chiral Carbon in Aliphatic StructuresĬhiral carbon present in a molecule can be identified in two steps as follows. For example, a molecule having two chiral carbons, essentially have four stereoisomers, two per each chiral carbon. Therefore, that kind of molecules can have more than two stereoisomers. Molecules having more than one chiral carbon atoms have two stereoisomers per one chiral carbon. sp or sp2 hybridized carbon atoms cannot be chiral because they cannot have four different groups around them due to the presence of π-bonds. Normally a chiral molecule contains at least one chiral carbon.
The carbon atom should always be sp 3 hybridized in order to be a chiral carbon. The chiral carbon is bonded to four different groups. A carbon atom can have a maximum of four bonds. Key Terms: Aliphatic Structure, Chirality, Chiral Carbon, Chiral Center, Ring Structure, StereoisomerismĪ chiral carbon is an asymmetric carbon. – Methods to Identify Chiral Carbons in Aliphatic Structure and Ring Structureģ. In organic chemistry, chiral centers are called chiral carbons. The chirality of a certain molecule is determined by the chiral centers present in that molecule. Chirality is the property of a molecule which says its mirror image is non-superimposable with the molecule. Unlike in structural isomerism, stereoisomerism includes the molecules having the same constitution of atoms but different spatial arrangements.

The concept of chirality comes under stereoisomerism. The two types of isomerism are structural isomerism and stereoisomerism. Isomerism is one of the major areas in organic chemistry which has a broad collection of molecules listed under it.
